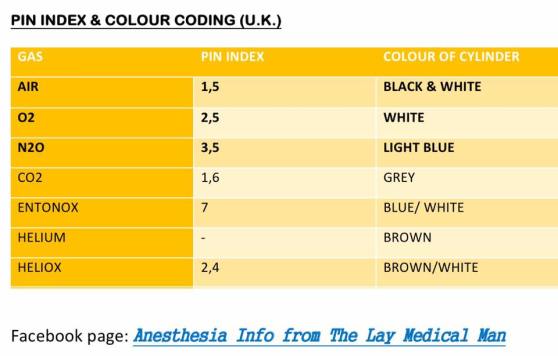
PIN INDEX



Inherited as Autosomal Recessive disease (MNEMO> Sickle Cell Disease causes Recession of RBC function)
A single DNA base change ( Beta chain) causes SCD
DNA base change is Adenine for Thymine & the resultant amino acid change is Valine for Glutamic Acid ( MNEMO> Adenine Added; Valine got a Welcome; Glutamine has to Go )
Thus Hb S is produced. As Valine is hydrophobic, the deoxygenated Hb is less water soluble and gets precipitated & polymerized inside the RBC
This polymerization slightly reduces the overall affinity for O2; otherwise the affinity for O2 is same for Hb A and Hb S
These changes also make the RBCs more rigid and contributes to sickling and microvascular occlusion
Regarding hypoxaemia, HbS will precipitate at a PO2 of 5–6 kPa (37-45 mm of Hg). As venous PO2 lies in this range, in case of homozygous individuals having only abnormal Hb will have continuous sickling
Patients with sickle cell trait experience sickling at much lower partial pressures (2.5–4 kPa / 19-30 mm of Hg )
Sickledex test produces a turbidity and becomes positive even with a very small amount of Hb S: so it CAN NOT differentiate between homo & heterozygous states
Reference: Smith T, Pinnock C, Lin T. Fundamentals of Anaesthesia, 3rd edn. Cambridge: Cambridge University Press, 2009; pp. 234–5
Tyrosine derived from thyroglobulin is combined with iodine to produce T3 & T4 (Thyroxin)
T3 is 5 times more active than T4, though T4 is produced in larger amounts
‘ACTIVITIES OF TSH’
Increase the size & number of thyroid gland cells
Increase iodide binding
Increase the release of thyroglobulin into the colloid of the gland
Increase pinocytosis of colloid by the thyroid cells
Increase hormone production
Increase release of already produced hormone from the bound thyroglobulin into the bloodstream
In bloodstream the hormones are 99% protein bound.
Thyroxin Binding Globulin (TBG) has the greatest affinity; but Albumin has the greatest capacity for binding the hormones. Thyroxine-binding prealbumin (TBPA) also bind them
REGULATION OF HORMONAL ACTIVITY
For regulation of the hormonal levels, the negative feedback is mediated by the unbound free fraction
Stress inhibits production
Warmth decreases production
Cold increases production
Glucocorticoids, dopamine & somatostatin inhibit TSH secretion
Reference: Smith T, Pinnock C, Lin T. Fundamentals of Anaesthesia, 3rd edn. Cambridge: Cambridge University Press, 2009; p. 474 .
Most effective for children <20 Kg (~ under 6 years of age) and for dermatomes below T10
Common side effects are weakness of legs, urinary retention etc
Because of this, sometimes a caudal block may necessitate overnight admission
The incidence of epidural hematoma has been reported as 1 in 80000 cases
Dose calculation can be done using Armitage ( 0.5 mL/kg for lumbosacral & 1 mL/kg for lumbar blockade, with 0.25% levobupivacaine ) or Scott formulas
Additives used in caudal block:
Preservative free Ketamine: Extend duration of analgesia; not used in infants <6 months of age due to fear of neurotoxicity
Clonidine : Extend duration of analgesia; not used in preterm infants and neonates due to higher incidence of bradycardia and apnoea. Provides postoperative sedation also.
Opioids when used as additives produce side effects like respiratory depression, pruritus & PONV
#EpiduralBlock , #Anaesthesia , #Anesthesia
References: De Beer DAH, Thomas ML. Caudal additives in children: solutions or problems? Br J Anaesth. 2003; 90: 487–498. Patel D. Epidural analgesia for children. Contin Educ Anaesth Crit Care Pain. 2006; 6(2): 63–66.
We ingest dietary iron in the form of either as free Fe or as haem bound Fe
The efficiency of absorption varies & is between 5-25% only
It also depends on total body Fe stores
We cannot expel iron by metabolism: then what’s the way? (1) We depend on slow losses through cell sloughing, menstruation, bleeding etc (2) We absorb only what we want
Fe is absorbed through duodenal & jejunal mucosa
If it’s free Fe, it attaches itself to a specific receptor ( it’s expression depends on the total body Fe stores) on the apical membrane of the cell. Fe is absorbed by active transport.
If haem-bound Fe, it enters the cell by pinocytosis, and inside the cell, haem is broken down
Here it binds with apoferritin to form ferritin, which is the intracellular storage form.
When required, Fe is released into the plasma. Here it binds with the transport molecule beta-transferrin ( it’s expression is also dependent on total body Fe store)
Carcinoid tumours are neuroendocrine tumours originating from enterochromaffin cells [GIT(~90%), gonads and bronchus mainly]
Some patients develop Carcinoid Syndrome, where the tumour secretes neuropeptides into systemic circulation
Usually they undergo firstpass metabolism in liver
If the patient is becoming symptomatic, due to the neuropeptide secretion, it’s either due to their production in large amounts to overwhelm the metabolic capacity of the liver or that they are released without going through the portal circulation
They secrete bio-active compounds like serotonin, histamine, catecholamines, bradykinin, kallikrein, substance-P, motilin etc
This can cause symptoms like bronchospasm, hypotension, hypertension, flushing etc
Pharmacologic treatment of intraoperative/ acute/ hemodynamic crises are with i.v. Octreotide, whereas for treatment of chronic symptoms, Somatostatin analogues like Lanreotide are used. Octreotide can also be used for prophylaxis. Should be continued postoperatively.
Vasoactive drugs like catecholamines and histamine releasing drugs like morphine, atracurium, succinylcholine, thiopentone etc should be avoided. Use of a test dose may reduce adverse events.
Antihistamines are also given prophylactically in case of gastric tumours
Another concern for the anesthesiologist in such patients is the possibility of Carcinoid heart disease. Here, the patient develops thickened valves resulting in tricuspid and pulmonary regurgitation and pulmonary stenosis (mitral and aortic insufficiency can also occur; but are less frequent). Pericarditis or myocardial metastases can also occur.
If we introduce nutritional support ( enteral or parenteral) based on the requirements of a regular healthy adult, to a malnourished patient, there will be a significant rise in basal insulin secretion, which will draw Potassium and Phosphate into the cell leading to hypokalemia, hypophosphatemia and fatal fluid shifts. ( Both rapid initiation and large amounts are dangerous). Phosphate depletion is also associated with increased urinary Magnesium excretion.
It can also be associated with Renal failure, Respiratory failure, Neuromuscular failure, Cardiac failure and Arrhythmias
This is known as “Refeeding Syndrome“
So to avoid this, in patients at risk ( e.g. chronic alcoholics, those who have not eaten anything in last 5 days etc) , we should introduce nutritional support at not more than 50% of the daily requirement , for the first two days.
Feeding rates can be increased to normal levels, if there is no evidence of refeeding syndrome clinically and biochemically, thereafter.
NICE guidelines for the high risk patients : start support with a maximum 10 kCal per kg per day, with thiamine & B complex supplementation. Biochemical parameters to be monitored closely.
There is no need for Prefeeding correction of electrolytes
#ICU , #nutrition , #NutritionInICU , #CriticalCare , #Anesthesia , #Anaesthesiology
Reference: Mehanna HM, Moledina J. Refeeding syndrome: What it is, and how to prevent and treat it. BMJ. 2008; 336(7659): 1495–1498
Hypotension , Hypoxemia and DIC are hallmarks (MNEMO> “AFE is Highly Dangerous”)
Hypotension & Fetal Distress occur in 100% of cases
DIC occur in 83% and indicate a bad prognosis
Cardiac arrest occur in around 87% of patients
Mortality is >60% ; it has been observed that only 15% survive with intact neurological function
Pulmonary Hypertension, CHF and DIC are key events in the pathogenesis
Pulmonary edema (occur in >90% of cases), Dyspnoea (occur in 49%) & Bronchospasm (occur in 15%) are the respiratory signs
Reference: Dedhia JD, Mushambi M. Amniotic fluid embolism. Contin Educ Anaesth Crit Care Pain. 2007; 7(5): 152–156. Gist RS, Stafford IP, Leibowitz AB et al. Amniotic fluid embolism. Anaesth Analg. 108(5): 1599–1602.