
ATRIAL NATRIURETIC FACTOR





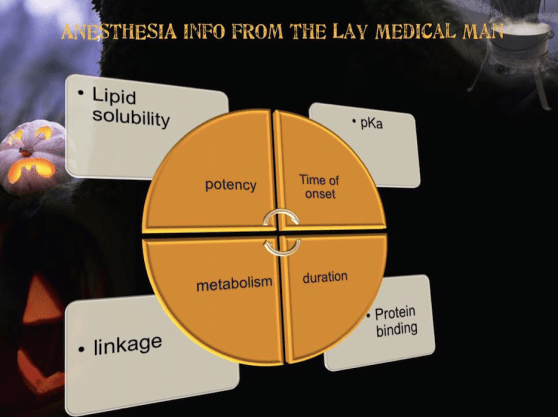
POTENCY:
Is affected by several factors including:
Hydrogen ion balance
Fiber size, type, and myelination
Vasodilator/vasoconstrictor properties (affects rate of vascular uptake)
Frequency of nerve stimulation
pH (acidic environment will antagonize the block)
Electrolyte concentrations (hypokalemia and hypercalcemia antagonizes blockade)
Lipid solubility.
DURATION OF ACTION
Is associated with lipid solubility.
Highly lipid soluble local anesthetics have a longer duration of action due to decreased clearance by localized blood flow and increased protein binding.
ONSET OF ACTION
Local anesthetics are weak bases and contain a higher ratio of ionized medication compared to non- ionized.
Increasing the concentration of non-ionized local anesthetic will speed onset.
In general, local anesthetics with a pKa that approximates physiologic pH have a higher concentration of non- ionized base resulting in a faster onset.
On the other hand, a local anesthetic with a pKa that is different from physiologic pH will have more ionized medication which slows onset.
For example, the pKa for lidocaine is 7.8 and 8.1 for bupivacaine. Lidocaine is closer to physiologic pH than bupivacaine. Lidocaine has a greater concentration on non-ionized local anesthetic than bupivacaine which results in a faster onset.
Non-ionized and ionized portions of local anesthetic solution exert distinct actions.
Lipid soluble, non-ionized form of the local anesthetic penetrates the neural sheath and membrane.
In the cell, the non-ionized and ionized forms equilibrate.
The ionized form of the local anesthetic binds with the sodium channel. Once “bound” to the sodium channel, impulses are not propagated along the nerve.
Clinically, onset of action is not the same for all local anesthetics with the same pKa. This is due to the intrinsic ability of the local anesthetic to diffuse through connective tissue.
So in general, local anesthetics with a pKa closest to the physiological pH generally have a higher concentration of non-ionized molecules and a more rapid onset.
Two notable exceptions are chloroprocaine and benzocaine. Chloroprocaine has a high pKa and rapid onset. Benzocaine does not exist in an ionized form and exerts its effects by alternate mechanisms.
Colorless, odourless, tasteless gas
Four times denser than air.
Density and viscosity are substantially higher than those of other inhalational anaesthetics.
Occurs in extremely low concentrations (0.0875 ppm) in the atmosphere, hence its name from the Greek ‘xenos’ meaning ‘stranger’.
Xenon has been used experimentally as an anaesthetic for more than 50 years
Recently there has been a renewed interest in xenon as a safe, effective and more environmentally friendly substitute for nitrous oxide (Sanders et al. 2003).
Manufactured by fractional distillation of liquefied air, currently at a cost of US $10 per litre (i.e. about 2,000 times the cost of producing N2O). This high cost is the major factor limiting its more widespread use, even when used in low-flow delivery systems.
Xenon has many of the properties of an ideal anaesthetic.
Its blood/gas partition coefficient (0.12) is lower than that of any other anaesthetic, giving rapid induction and emergence.
It is unlikely to be involved in any biochemical events in the body, and is not metabolised.
Xenon causes no significant changes in myocardial contractility, blood pressure or systemic vascular resistance, even in the presence of severe cardiac disease (Sanders et al. 2005).
The unique combination of analgesia, hypnosis, and lack of haemodynamic depression in one agent would make xenon a very attractive choice for patients with limited cardiovascular reserve
In contrast to other inhaled anaesthetic agents, xenon slows the respiratory rate and increases the tidal volume, thereby maintaining minute ventilation constant.
Airway pressure is increased during xenon anaesthesia, due to its higher density and viscosity rather than direct changes in airway resistance (Baumert et al 2002).
Because of its high cost xenon must be used in low-flow closed circuits. Crucial to this method of administration is accurate measurement of the concentration of xenon in the circuit. This measurement is generally difficult as xenon is diamagnetic and does not absorb infrared radiation (commonly used method to measure the concentrations of other agents), and its low reactivity precludes the use of specific fuel cell or electrode-type devices.
Xenon conducts heat better than other gases, and a technique based on thermal conductivity has proved to be effective (Luginbuhl et al 2002).
Because xenon is heavier than air, the speed of sound is slower in xenon than that in air, and this difference has been also been used to measure xenon concentration.
Because xenon is a normal constituent of the atmosphere, it does not add to atmospheric pollution when emitted from the anaesthesia circuit. This is in contrast to the other inhalational anaesthetics, which have ozone-depleting potential and pollute the atmosphere when released from the anaesthesia system (Marx et al. 2001).
On a molecular basis, N2O is 230 times more potent as a greenhouse gas than carbon dioxide. N2O released as a waste anaesthetic contributes roughly 0.1% of total global warming. The lifetime of N2O in the atmosphere is long—approximately 120 years.
The anaesthetic actions of xenon are thought to result primarily from noncompetitive inhibition of NMDA receptors (De Sousa et al. 2000), a property it shares with nitrous oxide.
In common with other NMDA receptor antagonists, xenon appears to have neuroprotective properties (Sanders et al. 2003).
Xenon is also an excellent analgesic, an action mediated by NMDA receptors (De Sousa et al. 2000).
Xenon also inhibits the plasma membrane Ca2+ pump, altering neuronal excitability and inhibiting the nociceptive responsiveness of spinal dorsal horn neurones.
#xenon ,#InhalationalAnaesthesia ,#GlobalWarming ,#Ozone , #OzoneDepletion ,#NobleGas ,#anaesthesia ,#ClinicalPharmacology,#pharmacology ,#research
(Reference : Jürgen Schüttler • Helmut Schwilden Modern Anesthetics ,Handbook of Experimental Pharmacology, vol 182)
CO2 is the most important stimulus for respiration
Receptors for CO2 are found in the medulla of the brain (central chemoreceptors)
Receptors for O2 are found mainly in carotid and aortic bodies
CO2 is the more important gas as the body has more capacity to store CO2 than O2 or hydrogen ions
In normal people at sea level, only 10% of the respiratory drive is due to hypoxic stimulation.
Unlike the central stimulation of hypercapnia, hypoxia causes central depression of the respiratory drive.
Acidosis (high H+ / low blood pH) stimulates respiration; conversely alkalosis depresses it.
For gas exchange, the lungs provide an interface of total surface area about 55 m2 via 700 million alveoli
Alveolar ventilation’ is that part of the total ventilation (i.e. all gas entering the lungs) that participates in gas exchange with pulmonary capillary blood; it is equal to total ventilation minus the ventilation of the conducting airways (i.e. dead-space ventilation).The average alveolar ventilation is about 4 L/min.
The alveolar–arterial oxygen gradient ( P(A-a)O2 ) is a measure of the oxygen that has reached the arterial blood supply as a ratio of the total oxygen in the alveoli. It is a useful index of pulmonary gas exchange function.
This requires that three elements are working correctly:
In a healthy individual breathing room air (at FiO2 0.21) the PO2 in alveolar air is 104 mmHg and in arterial blood 95 mmHg . PAO2 exceeds PaO2 by 15 mmHg .Thus, at an FiO2 of 21, the P(A–a)O2 is 15 mmHg
In blood, CO2 is present as:
Dissolved in blood plasma (5.3% in arterial blood)
Bound to haemoglobin as carbaminohaemoglobin within erythrocytes (4.5%)
In the form of bicarbonate attached to a base (90%)
Reference:”Understanding ABGs & Lung Function Tests” Muhunthan Thillai, Keith Hattotuwa
Drugs that act as Respiratory stimulants
Acetazolamide
Aminophyllines
Doxapram
Progesterone
Salicylates
Drugs that act as Respiratory depressants
Alcohol
Anaesthetics
Anticholinergics
Antihistamines
Barbiturates
Benzodiazepines
Opioids
MNEMO>
P͎A͎P͎A͎ C͎a͎r͎e͎s͎ M͎O͎R͎e͎ T͎H͎a͎n͎ N͎A͎T͎U͎R͎E͎
PAPAVERINE CODEINE MORPHINE THEBAINE
![]() ⭕️Oxygen content ( PaO2 ) is the pressure of oxygen molecules dissolved in blood, and is measured by ABG analysis with units of kPa or mmHg
⭕️Oxygen content ( PaO2 ) is the pressure of oxygen molecules dissolved in blood, and is measured by ABG analysis with units of kPa or mmHg
![]() ⭕️Oxygen saturation ( SaO2 ) is a measure of the percentage of haemoglobin sites that have oxygen bound, commonly measured with a pulse oximeter
⭕️Oxygen saturation ( SaO2 ) is a measure of the percentage of haemoglobin sites that have oxygen bound, commonly measured with a pulse oximeter
![]() ⭕️Oxygen content ( CaO2 ) is the real measure of blood oxygen quantity as it accounts for dissolved and haemoglobin bound oxygen. (i.e. CaO2 directly reflects the TOTAL number of oxygen molecules in arterial blood, both bound and unbound to hemoglobin. It is given as the volume of oxygen carried in each 100 ml blood (mL O 2 /100 mL). Normal CaO2 ranges from 16 to 22 ml O2/dl.
⭕️Oxygen content ( CaO2 ) is the real measure of blood oxygen quantity as it accounts for dissolved and haemoglobin bound oxygen. (i.e. CaO2 directly reflects the TOTAL number of oxygen molecules in arterial blood, both bound and unbound to hemoglobin. It is given as the volume of oxygen carried in each 100 ml blood (mL O 2 /100 mL). Normal CaO2 ranges from 16 to 22 ml O2/dl.
EXPLANATION:
![]() ⭕️Oxygen saturation ( SaO2 ) is expressed as the percentage of haemoglobin-binding sites that are occupied by oxygen, thereby forming oxyhaemoglobin.
⭕️Oxygen saturation ( SaO2 ) is expressed as the percentage of haemoglobin-binding sites that are occupied by oxygen, thereby forming oxyhaemoglobin.
![]() ⭕️Arterial blood is normally at 97–98% O 2 saturation (i.e. 98% of the available haemoglobin is combined with O 2 ), whereas venous blood is normally at 74% O2 saturation.
⭕️Arterial blood is normally at 97–98% O 2 saturation (i.e. 98% of the available haemoglobin is combined with O 2 ), whereas venous blood is normally at 74% O2 saturation.
![]() ⭕️O2 constitutes 21% of the atmosphere by volume and atmospheric PO2 is 159 mmHg at sea level . At an alveolar pressure of 104 mmHg, alveolar oxygen diffuses into pulmonary venous blood and raises its O2 content from 15 mL/100 mL to 20 mL/100 mL. Of this amount 19.75 mL is combined with haemoglobin and 0.25 mL is ‘free’ or dissolved in simple solution in the plasma. At this pressure of alveolar O2 , haemoglobin in the arterial blood normally becomes 98% saturated and and 2% of the haemoglobin remains reduced, i.e. free of oxygen.
⭕️O2 constitutes 21% of the atmosphere by volume and atmospheric PO2 is 159 mmHg at sea level . At an alveolar pressure of 104 mmHg, alveolar oxygen diffuses into pulmonary venous blood and raises its O2 content from 15 mL/100 mL to 20 mL/100 mL. Of this amount 19.75 mL is combined with haemoglobin and 0.25 mL is ‘free’ or dissolved in simple solution in the plasma. At this pressure of alveolar O2 , haemoglobin in the arterial blood normally becomes 98% saturated and and 2% of the haemoglobin remains reduced, i.e. free of oxygen.
![]() ⭕️PaO2 is determined by alveolar PO2 and the state of the alveolar-capillary interface, not by the amount of hemoglobin available to soak them up. PaO2 is not a function of hemoglobin content or of its characteristics. This explains why, for example, patients with severe anemia or carbon monoxide poisoning or methemoglobinemia can (and often do) have a normal PaO2.
⭕️PaO2 is determined by alveolar PO2 and the state of the alveolar-capillary interface, not by the amount of hemoglobin available to soak them up. PaO2 is not a function of hemoglobin content or of its characteristics. This explains why, for example, patients with severe anemia or carbon monoxide poisoning or methemoglobinemia can (and often do) have a normal PaO2.
![]() ⭕️The most common physiologic disturbance of lung architecture, and hence of a reduced PaO2, is ventilation-perfusion (V-Q) imbalance. Less common causes are reduced alveolar ventilation, diffusion block, and anatomic right to left shunting of blood.
⭕️The most common physiologic disturbance of lung architecture, and hence of a reduced PaO2, is ventilation-perfusion (V-Q) imbalance. Less common causes are reduced alveolar ventilation, diffusion block, and anatomic right to left shunting of blood.
![]() ⭕️Think of PaO2 as the driving pressure for oxygen molecules entering the red blood cell and chemically binding to hemoglobin; the higher the PaO2, the higher the SaO2.
⭕️Think of PaO2 as the driving pressure for oxygen molecules entering the red blood cell and chemically binding to hemoglobin; the higher the PaO2, the higher the SaO2.
![]() ⭕️ In contrast to the other two variables, CaO2 depends on the hemoglobin content and is directly related to it; Since the dissolved oxygen contributes minimally to CaO2 under physiologic conditions, CaO2 is determined almost entirely by hemoglobin content and SaO2, and is related linearly to either variable.
⭕️ In contrast to the other two variables, CaO2 depends on the hemoglobin content and is directly related to it; Since the dissolved oxygen contributes minimally to CaO2 under physiologic conditions, CaO2 is determined almost entirely by hemoglobin content and SaO2, and is related linearly to either variable.
CaO2 = Hb (gm/dl) x 1.34 ml O2/gm Hb x SaO2 + PaO2 x (.003 ml O2/mm Hg/dl).

Comes as 5 mg/mL ampoules
Blocks α, β1, and β2 adrenergic receptor sites.
Decreases heart rate and peripheral vascular resistance.
Ratio of alpha to beta blockade depends upon the route of administration (1:3 oral versus 1:7 IV)
Onset of action: 2‐5 minutes
Will not cause bronchoconstriction
Duration: 2‐4 hours
IV Bolus: 20 mg over at least 2 minutes as initial dose, may repeat with doses of 40-‐ 80 mg q10min; Do not exceed total dose of 300 mg
Infusion [ dilution 1 mg/mL] : starting 2 mg/min (2 mL/min) – 8 mg/min titrated to response. Do not exceed total dose of 300
As cumulative dose nears 300mg IV, duration of action extends to nearly 18 hours.
Ref: B Xu, F Charlton, A Makris, A Hennessy – Journal of hypertension, 2014