Category Archives: Exams EDAIC FRCA
THE SPECIFIC MECHANISMS OF ACTION OF #MANNITOL IN VARIOUS CLINICAL SITUATIONS
Mannitol is a monosaccharide available as 10% & 20% solutions
DURING NEUROSURGERY/ IN NEUROCRITICAL CARE:
✔️Mannitol is freely filtered in the glomerulus but won’t get reabsorbed in the tubules; so it will drive water from the interstitium which gets eliminated as urine. Hence acts as an osmotic diuretic
✔️When blood brain barrier is intact, the osmotic gradient created by mannitol will move water from the cerebral extravascular compartment to the intravascular space, reducing ICP. If blood brain barrier is not intact, it will worsen cerebral edema.
✔️The expansion of the plasma volume caused by mannitol will reduce the viscosity and improve cerebrovascular microcirculation and oxygenation. The increase in cardiac output can also cause an increase in regional blood flow which will cause a compensatory cerebrovascular vasoconstriction in areas where autoregulation is intact.
IN CRUSH INJURY / MYOGLOBINURIA
✔️Will release renal prostaglandins, which will cause renal vasodilation and increase tubular urine flow causing a solute washout and avoidance of tubular obstruction #TheLayMedicalMan
MECHANISM BEHIND ADVERSE EFFECTS
✔️The initial increase in plasma volume as a result of drawing of water into the vascular component and the resultant increase in cardiac output can precipitate heart failure in cardiac patients
✔️The osmotic diuresis can cause hypernatremia [increases urinary losses of both sodium and electrolyte-free water] , metabolic acidosis and hyperosmolarity. It has been advised that therapy should be monitored and titrated so that osmolarity doesn’t go up beyond 300 mOsm/L
✔️The rise in the plasma potassium concentration following hypertonic mannitol is due to the movement of potassium out of the cells into the extracellular fluid as the rise in cell potassium concentration induced by water loss favors passive potassium exit through potassium channels in the cell membrane
✔️Though it has been used for renal protection, the reduction in renal perfusion resulting from hypovolemia caused by diuresis can adversely affect renal function; so should be avoided in patients with renal dysfunction
#Neuroanesthesia , #Anesthesia , #Neurology , #CriticalCare
VIVA SCENE: DESCRIPTION OF THE IMPORTANT EVENTS IN THE CARDIAC CYCLE SYNCHRONISED WITH THE EKG & VENOUS PRESSURE WAVE FORMS
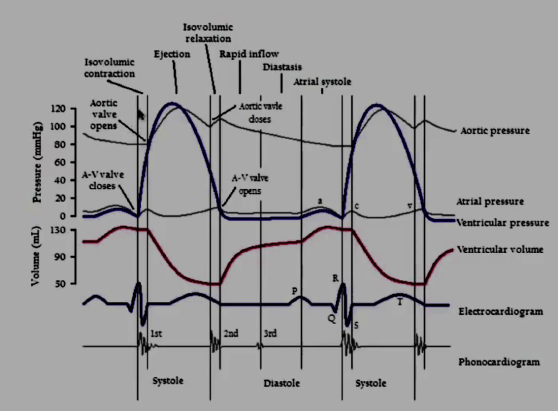
1. ATRIAL CONTRACTION (Phase 1): It is initiated by the P wave of the ECG which represents electrical depolarization of the atria. Atrial contraction does produce a small increase in venous pressure that can be noted as the “a-wave“. Just following the peak of the a-wave is the x-descent. Atrial contraction normally accounts ONLY for about 10% of left ventricular filling when a person is at rest. At high heart rates when there is less time for passive ventricular filling, the atrial contraction may account for up to 40% of ventricular filling. This is sometimes referred to as the “atrial kick.” The atrial contribution to ventricular filling varies inversely with duration of ventricular diastole and directly with atrial contractility. The volume of blood at the end of the filling phase is the end diastolic volume and is around 120 mL in the adult. S4 sound is caused by vibration of the ventricular wall during atrial contraction. Generally, it is noted when the ventricle compliance is reduced (“stiff” ventricle) as occurs in ventricular hypertrophy and in many older individuals.
2. Isovolumetric Contraction (Phase 2): This phase of the cardiac cycle begins with the appearance of the QRS complex of the ECG, which represents ventricular depolarization. The AV valves close when intraventricular pressure exceeds atrial pressure. Closure of the AV valves results in the first heart sound (S1). During the time period between the closure of the AV valves and the opening of the aortic and pulmonic valves, ventricular pressure rises rapidly without a change in ventricular volume (i.e., no ejection occurs). Ventricular volume does not change because all valves are closed during this phase. Contraction, therefore, is said to be isovolumetric. The “c-wave” noted in the venous pressure may be due to bulging of A-V valve leaflets back into the atria. Just after the peak of the c wave is the y’-descent.
3. Rapid Ejection (Phase 3): Ejection begins when the intraventricular pressures exceed the pressures within the aorta (~80 mm of Hg) and pulmonary artery, which causes the aortic and pulmonic valves to open. Left atrial pressure initially decreases as the atrial base is pulled downward, expanding the atrial chamber. Blood continues to flow into the atria from their respective venous inflow tracts and the atrial pressures begin to rise. This rise in pressure continues until the AV valves open at the end of phase 5.
4. Reduced Ejection (Phase 4): Approximately 200 msec after the QRS and the beginning of ventricular contraction, ventricular repolarization occurs as shown by the T-wave of the electrocardiogram. Repolarization leads to a decline in ventricular active tension and pressure generation; therefore, the rate of ejection (ventricular emptying) falls. Ventricular pressure falls slightly below outflow tract pressure; however, outward flow still occurs due to kinetic (or inertial) energy of the blood. Left atrial and right atrial pressures gradually rise due to continued venous return from the lungs and from the systemic circulation, respectively.
5. Isovolumetric Relaxation (Phase 5): When the intraventricular pressures fall sufficiently at the end of phase 4, the aortic and pulmonic valves abruptly close (aortic precedes pulmonic) causing the second heart sound (S2) and the beginning of isovolumetric relaxation. Valve closure is associated with a small backflow of blood into the ventricles and a characteristic notch (incisura or dicrotic notch) in the aortic and pulmonary artery pressure tracings. Although ventricular pressures decrease during this phase, volumes do not change because all valves are closed. The volume of blood that remains in a ventricle is called the end-systolic volume and is ~50 ml in the left ventricle. The difference between the end-diastolic volume and the end-systolic volume is ~70 ml and represents the stroke volume. Left atrial pressure (LAP) continues to rise because of venous return from the lungs. During isovolumetric ventricular relaxation, atrial pressure rises to 5 mmHg in the left atrium and 2 mmHg in the right atrium.
6. Rapid Filling (Phase 6): As the ventricles continue to relax at the end of phase 5, the intraventricular pressures will at some point fall below their respective atrial pressures. When this occurs, the AV valves rapidly open and passive ventricular filling begins. The opening of the mitral valve causes a rapid fall in LAP. The peak of the LAP just before the valve opens is represented by the “v-wave.” This is followed by the y-descent of the LAP. A similar wave and descent are found in the right atrium and in the jugular vein. When a third heart sound (S3) is audible during rapid ventricular filling, it is often pathological and is caused by ventricular dilatation.
7. Reduced Filling (Phase 7): As the ventricles continue to fill with blood and expand, they become less compliant and the intraventricular pressures rise. The increase in intraventricular pressure reduces the pressure gradient across the AV valves so that the rate of filling falls late in diastole. In normal, resting hearts, the ventricle is about 90% filled by the end of this phase. In other words, about 90% of ventricular filling occurs before atrial contraction (phase 1) and therefore is passive.
8. Right Vs Left: The major difference between the right and left side of the cardiac chambers, is that the peak systolic pressures of the right heart are substantially lower than those of the left heart, and this is because pulmonary vascular resistance is lower than systemic vascular resistance. Typical pulmonary systolic and diastolic pressures are 24 and 8 mm Hg, respectively.
9. Jugular Venous Pressure Summary: Right atrial pressure pulsations are transmitted to jugular veins. Thus, atrial contractions produce the first pressure peak called the a wave. Shortly thereafter, the second peak pressure called the c wave follows and this is caused by the bulging of the tricuspid valve into the right atrium. After the c wave, the right atrial pressure decreases (‘x’ descent) because the atrium relaxes and the tricuspid valve descends during ventricular emptying. As the central veins and the right atrium fill behind a closed tricuspid valve, the right atrial pressure rises towards a third peak, the v wave. When the tricuspid valve opens at the end of ventricular systole, right atrial pressure decreases again as blood enters the relaxed right ventricle (‘y’ descent). The right atrial pressure begins to rise shortly as blood returns to the right atrium and the right ventricle together during diastole.
 Ref: Principles of Physiology for the Anaesthetist, Peter Kam, Ian Power, http://www.cvphysiology. com
Ref: Principles of Physiology for the Anaesthetist, Peter Kam, Ian Power, http://www.cvphysiology. com
#CardiacCycle , #Physiology , #Anesthesia
CO2 ABSORBENTS
WHAT IS POYNTING EFFECT ❓
#SCAVENGING IN #ANESTHESIA
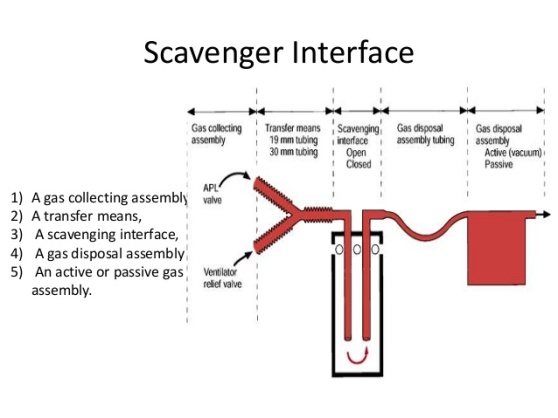 Scavenging refers to the method of extracting waste gases from the breathing system and venting them to an area where they will not be directly inhaled by staff or other patients.
Scavenging refers to the method of extracting waste gases from the breathing system and venting them to an area where they will not be directly inhaled by staff or other patients.
- Scavenging systems can be classified as open or closed.
- Open refers to the basic system of extracting the gas from its point of entry into the theatre
- Closed systems are more common and can be further subdivided into active and passive
- No conservation of volatile agent is possible with either the active or the passive systems; conservation must occur within the anaesthetic breathing system itself, by the use of a circle system and low-flow anaesthesia.
- The active and the passive both pass any waste gas to the atmosphere, polluting it to the same extent.
- In the passive scavenging system an exhaust port collects the waste gases from the expiratory valve of the breathing system or from the ventilator and the gases pass through the transfer system (which consists of 30 mm low-resistance tubing) to the outside of the building, preferably above roof level.
- If the theatre air is not recirculated, the waste gases can be piped to the exit port of the theatre ventilation system.
- In the passive system, the gases are pushed to the atmosphere solely by the expiratory power of the patient
- If the pathway to the atmosphere involves a vertical passage of gas, then the patient must overcome the atmospheric pressure required to push the gas over this distance; may be several floors of hospital! Means significant forces has to be overcome.
- The use of gases with higher density, like nitrous oxide, adverse atmospheric conditions like high winds etc, will further increase the forces required to expel waste gases; this can even affect the cardiopulmonary status of the patient.
#EPINEPHRINE ( #Adrenaline) : Pharmacological Highlights
🔻Epinephrine is an agonist of alpha 1, β1 , and β2 adrenoceptors. An intravenous infusion of epinephrine produces an increase in mean arterial pressure (MAP) characterized by selectively enhanced systolic pressure with no change in diastolic pressure.
🔻Epinephrine exerts positive chronotropic and inotropic actions by stimulation of β1 adrenoceptors
🔻Epinephrine also increases the rate of myocardial relaxation and enhances early LV filling, thereby improving diastolic function. These combined effects result in a dramatic increase in cardiac output.
🔻Epinephrine (0.01–0.03 ug kg –1 min –1 ) has been shown to produce similar hemodynamic effects with less pronounced tachycardia than dobutamine (2.5–5.0 ug kg–1 min–1 ) in patients after coronary artery bypass graft (CABG) surgery
🔻Predictable increase in cardiac output, favours the use of epinephrine as the primary inotropic drug for the management of LV dysfunction after cardiopulmonary bypass
🔻Epinephrine causes direct positive dromotropic effects ( leading to increase in conduction velocity and reduction of the refractory period of the AV node, His bundle, Purkinje fibers, and ventricular muscle)
🔻This may contribute to detrimental increases in ventricular rate in patients with atrial flutter or fibrillation and the occurrence of ventricular arrhythmias
🔻The overall effect of epinephrine on blood flow to a specific organ depends on the relative balance of alpha 1 and β2 adrenoceptors located in the vasculature.
🔻β2 -Adrenoceptors are sensitive to lower doses of epinephrine and, as a result, peripheral vasodilation and modest reductions in arterial pressure are observed with such doses
🔻In contrast, the effects of epinephrine on alpha 1 -adrenoceptors predominate at greater doses with marked increases in systemic vascular resistance and arterial pressure.
🔻The intense vasoconstriction produced by high doses of epinephrine may adversely impede LV ejection by increasing after load after cardiopulmonary bypass. Thus, greater doses of epinephrine may be used in combination with arterial vasodilators such as sodium nitroprusside to optimize contractile performance in such situations .
🔻Adrenaline via alpha 1 receptors also mediates (1) venoconstriction & enhanced venous return (2) Pulmonary vasoconstriction and increases in pulmonary arterial pressures.
🔻 Pre-existing β-blockade by nonselective β-blocker propranolol abolishes the decrease in systemic vascular resistance from epinephrine-induced stimulation of β2 adrenoceptors and potentiates peripheral vasoconstriction mediated by unopposed alpha 1 adrenoceptors.
🔻The positive inotropic and chronotropic effects of epinephrine are also attenuated in the presence of pre-existing β-blockade and greater doses of epinephrine are required to overcome this competitive blockade
🔻Complete pharmacologic blockade of β1 and β2 adrenoceptors may theoretically make the hemodynamic effects of epinephrine indistinguishable from those of the pure alpha 1 adrenoceptor agonist phenylephrine.
#NorAdrenaline , #CriticalCare , #vasopressors , #TheLayMedicalMan , #IntensiveCare , #Pharmacology, #anesthesia , #anaesthesia, #drugs
(Reference: Paul S. Pagel and David C. Warltier, Essential drugs in anesthesia practice Positive inotropic drugs, Anesthetic Pharmacology, 2nd edition)
JNC 8 GUIDELINES FOR MANAGEMENT OF SYSTEMIC HYPERTENSION
(1)📌In the general population aged 60 years or more , initiate pharmacologic treatment to lower blood pressure (BP) at systolic blood pressure (SBP) ≥150 mm Hg or diastolic blood pressure (DBP) ≥90 mm Hg and treat to a goal SBP <150 mm Hg and goal DBP <90 mm Hg
(2)📌In the general population aged ≥60 years, if pharmacologic treatment for high BP results in lower achieved SBP (eg, <140 mm Hg) and treatment is well tolerated and without adverse effects on health or quality of life, treatment does not need to be adjusted.
(3)📌In the general population <60 years, initiate pharmacologic treatment
(a) to lower BP at DBP ≥90mmHg and treat to a goal DBP <90mmHg.
(b) to lower BP at SBP ≥140 mm Hg and treat to a goal SBP <140 mm Hg.
(4)📌In the population aged ≥18 years with (i) diabetes & (ii) chronic kidney disease (CKD), initiate pharmacologic treatment to lower BP at SBP ≥140 mmHg or DBP ≥90 mmHg and treat to goal SBP <140mmHg and goal DBP <90mmHg.
(5)📌In the general nonblack population, including those with diabetes, initial antihypertensive treatment should include a thiazide-type diuretic, calcium channel blocker (CCB), angiotensin-converting enzyme inhibitor
(ACEI), or angiotensin receptor blocker (ARB).
(6)📌In the general black population, including those with diabetes, initial antihypertensive treatment should include a thiazide-type diuretic or CCB.
(7)📌In the population aged 18 years with CKD, initial (or add-on) antihypertensive treatment should include an ACEI or ARB to improve kidney outcomes. This applies to all CKD patients with hypertension regardless of race
or diabetes status.
(8)📌If goal BP is not reached within a month of treatment, increase the dose of the initial drug or add a second drug from one of the classes : thiazide-type diuretic, CCB,ACEI, or ARB. The clinician should continue to assess BP and adjust the treatment regimen until goal BP is reached.
(9)📌If goal BP cannot be reached with 2 drugs, add and titrate a third drug from the list mentioned above (). Do not use an ACEI and an ARB together in the same patient.
(10)📌If goal BP cannot be reached using only the drugs mentioned above, because of a contraindication or the need to use more than 3 drugs to reach goal BP, antihypertensive drugs from other classes can be used.
#hypertension , #medicine , #TheLayMedicalMan , #jnc8 , #HTN , #anesthesia , #pharmacology , #BloodPressure ,#BP
NORADRENALINE: PHARMACOLOGICAL HIGHLIGHTS & COMPARISON WITH ADRENALINE
Noradrenaline (Norepinephrine) is a directly and indirectly acting sympathomimetic amine which stimulates alpha 1 and β1 adrenoceptors, but, in contrast to Adrenaline (epinephrine), has little effect on β2 adrenoceptors.
🖍These actions produce positive inotropic effects, intense vasoconstriction, increases in arterial pressure, and relative maintenance of cardiac output.
🖍Noradrenaline increases arterial pressure while simultaneously enhancing contractile state and venous return by reductions in venous capacitance, thereby augmenting stroke volume and ejection fraction. In contrast, pure alpha 1 adrenoceptor agonists such as phenylephrine and methoxamine further compromise cardiac output in failing myocardium and contribute to peripheral hypoperfusion despite an increase in arterial pressure.
🖍In contrast to adrenaline, noradrenaline does not substantially affect heart rate because activation of baroreceptor reflexes resulting from arterial vasoconstriction usually counteracts β1 mediated, direct, positive, chronotropic effects.
🖍Its arrhythmogenic potential is considerably less than that of adrenaline. Thus, substitution of noradrenaline for adrenaline may be appropriate in the therapeutic management of cardiogenic shock when atrial or ventricular arrhythmias are present.
🖍Intravenous infusions of noradrenaline (0.03–0.90 mg kg –1 per minute) have been shown to increase arterial pressure, LV stroke work index, cardiac index, and urine output in septic patients with hypotension that was unresponsive to volume administration, dopamine or dobutamine
🖍Causes relatively greater increases in systemic vascular resistance and diastolic arterial pressure than adrenaline.
🖍The drug has a duration of action of 30–40 minutes; tachyphylaxis occurs with prolonged administration.
🖍The drug produces coronary vasodilatation, leading to a marked increase in coronary blood flow. However, as myocardial work may increase, the balance of myocardial oxygen consumption and delivery may lead to ischaemia on noradrenaline.
🖍Reflex vagal stimulation leads to a compensatory bradycardia
🖍The cerebral blood flow and oxygen consumption are decreased by the administration of noradrenaline; mydriasis also occurs
🖍The glomerular filtration rate is usually well maintained with noradrenaline; but it decreases the renal blood flow and this represents a major limitation on the prolonged use of high doses of norepinephrine.
🖍Noradrenaline increases the contractility of the pregnant uterus; this may lead to fetal bradycardia and asphyxia
🖍Noradrenaline may decrease insulin secretion, leading to hyperglycaemia
🖍The drug is pharmaceutically incompatible with barbiturates and sodium bicarbonate
(Reference: Paul S. Pagel and David C. Warltier, Essential drugs in anesthesia practice Positive inotropic drugs, Anesthetic Pharmacology, 2nd edition)

VIVA AID: BISPECTRAL INDEX
💆The EEG bispectrum is a high-order statistical computation derived from the analog EEG.
💆The BIS is a combination of three weighted parameters: (i) the burst suppression ratio (the proportion of isoelectric EEG signal in an epoch); (ii) the beta ratio (a measure of the proportion of signal power in the high vs medium frequency range); and (iii) the SynchFastSlow (relative synchrony of fast and slow waves)
💆Changes in frequency and power alone ( as done with conventional power spectral analysis) have been shown to be inconsistent when attempting to measure anesthetic depth.
💆Bispectral analysis incorporates information on power and frequency with the phase coupling information that is more indicative of anesthetic depth but not present in other clinical applications of EEG.
💆The BIS uses a combination of EEG subparameters that were selected after analysis of a large database of EEGs to demonstrate specific ranges for varying phases of anesthetic effect
💆These parameters were then combined to form the optimum configuration for monitoring of the hypnotic state.
💆The BIS is then displayed as a dimensionless number between 0 and 100 with the lower numbers corresponding to deeper levels of hypnosis.
💆There are normal, genetically determined low-voltage EEG variants among the population that can result in abnormally low BIS values in awake patients; therefore, it is important to obtain baseline values before the induction of anesthesia
💆BIS is not able to predict movement in response to surgical stimulation because the generation of reflexes is likely to be at spinal cord rather than cortical level
💆BIS does not fully reflect the synergistic effect of opioids with hypnotic agents
💆The presence of electromyographic artefacts, poor signal quality, and electrical artefacts such as those from electro-cautery and forced air warming units can cause spurious values to be displayed by the BIS monitor.
💆With the administration of ketamine, the BIS may remain high, possibly due to the excitatory actions of ketamine, and, therefore, the BIS monitor is not reliable when used to monitor hypnosis with ketamine.
💆There have been studies in which the BIS monitor has not been shown to reflect the hypnotic contribution to the anesthetic by nitrous oxide.
💆Potential benefits from the routine use of the BIS monitor include
➖decreased risk of awareness
➖improved titration of anesthetic agents and
➖decreased recovery room time
💆The BIS also gives the anesthetist additional information to consider when selecting drugs for interventions, for example, when making the decision whether to deepen anesthesia with a volatile agent, add more analgesia with an opioid, or use a vasoactive drug.
💆Also note:
➖The BIS may drop after giving a neuromuscular blocking agent if excessive EMG was present prior to giving it.
➖Ischemia attenuates the amplitude and frequency of the EEG signal, which may result in a decrease in BIS
➖Hypothermia decreases brain activity, and may decrease BIS
➖Muscle shivering, tightening, twitching etc may increase EMG and increase BIS
➖Artifacts in the higher frequency ranges [e.g. use of any mechanical device that could generate high frequency activity like patient warmer]can artificially increase the BIS value
➖Is the BIS decreasing when you think it should be increasing? Think of Paradoxical Delta pattern (characterized by a pronounced slowing of the EEG) which occurs over a short period of time (2-3 minutes).
➖If the sensor is placed over the temporal artery, pulse artifacts can cause the BIS value to be inappropriately low. Check EEG waveform for presence of pulse artifacts and move sensor if necessary.
➖Blinking or rolling his/her head by the patient, may cause artifacts that mimic slow frequency EEG patterns.
Reference: The BIS monitor: A review and technology assessment, James W. Bard, AANA Journal/December 2001/Vol. 69, No. 6